Archive for the ‘Consumerism’ Category
We May Be Leaving The EU But The Soviet State Of Britain Is Already Here.
Email to my MP, Sir Oliver Letwin, concerning the Investigatory Powers Bill
From: Peter Reynolds
Sent: 25 November 2016 15:01
To: Oliver Letwin
Subject: The Investigatory Powers Bill
Dear Oliver,
Perhaps you can explain to me on what basis of reason, justice or liberty the following organisations will shortly have access at will to my (and your) internet browsing history?
This is an outrageous intrusion into my private life and completely unacceptable. I think it’s time we started a UK campaign for the right to bear arms. Every day it seems the government is becoming more and more of an oppressor. I reject this entirely and it does not have my consent.
Metropolitan Police Service
City of London Police
Police forces maintained under section 2 of the Police Act 1996
Police Service of Scotland
Police Service of Northern Ireland
British Transport Police
Ministry of Defence Police
Royal Navy Police
Royal Military Police
Royal Air Force Police
Security Service
Secret Intelligence Service
GCHQ
Ministry of Defence
Department of Health
Home Office
Ministry of Justice
National Crime Agency
HM Revenue & Customs
Department for Transport
Department for Work and Pensions
NHS trusts and foundation trusts in England that provide ambulance services
Common Services Agency for the Scottish Health Service
Competition and Markets Authority
Criminal Cases Review Commission
Department for Communities in Northern Ireland
Department for the Economy in Northern Ireland
Department of Justice in Northern Ireland
Financial Conduct Authority
Fire and rescue authorities under the Fire and Rescue Services Act 2004
Food Standards Agency
Food Standards Scotland
Gambling Commission
Gangmasters and Labour Abuse Authority
Health and Safety Executive
Independent Police Complaints Commissioner
Information Commissioner
NHS Business Services Authority
Northern Ireland Ambulance Service Health and Social Care Trust
Northern Ireland Fire and Rescue Service Board
Northern Ireland Health and Social Care Regional Business Services Organisation
Office of Communications
Office of the Police Ombudsman for Northern Ireland
Police Investigations and Review Commissioner
Scottish Ambulance Service Board
Scottish Criminal Cases Review Commission
Serious Fraud Office
Welsh Ambulance Services National Health Service Trust
Seriously, how can any MP with any integrity or honour vote for such Soviet-style government snooping?
Kind regards,
Peter Reynolds
Cruel And Irresponsible Response from UK Government To Parliamentary Report On Medicinal Cannabis.
Unsurprisingly perhaps, the response to the recent call from MPs and peers to legalise cannabis for medicinal use has come straight from the top. Theresa May’s longstanding reputation as a denier of science and evidence on drugs policy is reinforced by her peremptory dismissal of the expert report. It seems that, at least in the short term, the UK government is sticking by a policy that is discredited, ridiculous and deeply cruel.
It fell to Sarah Newton MP, minister of state at the Home Office, to respond to a parliamentary question from Roger Godsiff, Labour MP for Birmingham, Hall Green.
“To ask the Secretary of State for the Home Department, if she will respond to the recommendations of the report by the All-Party Parliamentary Group for Drug Policy Reform Accessing Medicinal Cannabis: Meeting Patients’ Needs, published in September 2016.”
“The Prime Minister responded to the All-Party Parliamentary Group for Drug Policy Reform’s report ‘Accessing Medicinal Cannabis: Meeting Patients’ Needs’ on the 27 October.
Cannabis is controlled as a Class B drug under the Misuse of Drugs Act 1971 and, in its raw form, currently has no recognised medicinal benefits in the UK. It is therefore listed as a Schedule 1 drug under the Misuse of Drugs Regulations 2001.
It is important that all medicines containing controlled drugs are thoroughly trialled to ensure they meet rigorous standards so that doctors and patients are sure of their efficacy and safety. To do otherwise for cannabis would amount to a circumvention of the clearly established and necessary regime for approving medicines in the UK.”
In other words, this is nothing more than a re-statement of the same position that the UK government has held since 1971 when legal access to medicinal cannabis was halted. Quite clearly the government has given no consideration at all to the vast amount of scientific evidence and international experience that has accumulated over the last 45 years. The latest report which took nine months to produce, took evidence from over 600 witnesses and included a review of over 20,000 scientific studies is simply cast aside. To be honest, I doubt whether it has even been read by Ms May or anyone in the Home Office or Department of Health. This is the standard that now prevails in the UK – government of the people by an unaccountable, out-of-touch, unresponsive cabal of individuals elected by a deeply flawed system that gives democracy a bad name.
On the face of it, the claim that all medicines must be thoroughly trialled seems plausible – but it is not. It is a misleading half-truth clearly intended to squash the call for access to medicinal cannabis by painting a false picture.
Doctors are allowed to prescribe any medicine, licensed or unlicensed, as they see fit, based on their own judgement. But prescribing of cannabis is specifically prohibited by Statutory Instrument despite the scientific consensus that it is far less dangerous than many, probably most commonly prescribed medicines.
So it’s not a level playing field. It’s a policy that is based on prejudice and scaremongering about recreational use of cannabis. Ms Newton’s answer is at best disingenuous but then she probably doesn’t even realise that herself. For many years Home Office policy has been systematically to mislead and misinform on cannabis and evidently under Ms May’s successor, Amber Rudd MP, such dishonesty continues.
Something will eventually force the government’s hand to change its absurd position on cannabis. Sadly the very last consideration will be scientific evidence or the will of the people. Such factors hold no sway with UK governments. Only when enough of the political elite open their eyes and examine their conscience, or some key individuals or their family members, experience the need for medicinal cannabis will change become possible. Alternatively, political upheaval may present an opportunity. The Liberal Democrats were too cowardly, weak and concerned with building their personal careers when in coalition to advance the cause they now so bravely advocate. Perhaps the SNP, with 56 MPs, all in favour of medicinal cannabis may be our best hope.
Sarah Newton is merely a puppet of the Home Office bureaucracy. Theresa May’s mendacious position on all aspects of drugs policy is well established and she is as stubborn and bigoted as they come on such matters. Only when she, in person, is subject to sufficient pressure will this cruel, ignorant and hateful policy change.
CLEAR Statement Concerning Cannabis Legalisation Measures In US Election.
“This is marvellous news for liberty, health and human rights. The USA, unlike Britain, has a functioning democracy where the will of the people prevails rather than the bigotry and self-interest of politicians. It is wonderful to see that truth, justice and evidence is winning out over the lies and misinformation we have been fed about cannabis for almost 100 years.
In 1971, the British government abdicated all responsibility on cannabis and abandoned our communities and our children to criminal gangs. Since then all the harms have multiplied exponentially. The laws against cannabis fund organised crime, promote dangerous hidden farms which are fire risks, the destruction of rental property, selling to children, contaminated ‘moonshine’ cannabis, gang violence, lives ruined by criminal records and the cruel denial of safe, effective medicine that can relieve pain, suffering and disability.
Donald Trump has supported access to medicinal cannabis all along. Many British politicians who consider him to be an unreasonable person should now look to themselves and ask whether they are being reasonable by supporting prohibition, even for medical use.
It is time for Theresa May, Amber Rudd and the UK government to take responsibility for the £6 billion pa cannabis market. The tide of legalisation is now unstoppable and it would be deeply irresponsible for them to fail to act. They must grasp this nettle now!”
Peter Reynolds, president of CLEAR Cannabis Law Reform
MHRA Backtracking Super Fast On CBD Ban.
 In advance of the meeting between the UK Cannabis Trade Association (UKCTA) and the MHRA on Thursday, there has been a flurry of activity which amounts to a climb down by the regulator.
In advance of the meeting between the UK Cannabis Trade Association (UKCTA) and the MHRA on Thursday, there has been a flurry of activity which amounts to a climb down by the regulator.
This statement was published on the MHRA website at lunchtime today.
“Update 1 November 2016
An MHRA spokesperson said:
While MHRA has given its opinion that products containing cannabidiol (CBD) used for medical purposes are medicines, we have also carefully considered the needs of individuals using CBD products to treat or manage the symptoms of medical conditions.
Our primary concern is patient safety. In order to ensure that products remain available until individuals have the opportunity to discuss their treatment with their doctor, companies now have until 31 December 2016 to voluntarily operate within the law, by withdrawing their existing products from the market, or working with MHRA to satisfy the legal requirements of the Human Medicines Regulations 2012.
We have today written to the manufacturers of CBD to make them aware of the timeline for engagement.
It is vital that medicines meet safety, quality and efficacy standards to protect public health.”
Originally the MHRA wrote to CBD suppliers in threatening terms:
“You must cease to sell, supply, promote, advertise or process orders for the above products until appropriate authorisation has been granted for them. You must confirm this in writing within 28 days from the date of this letter that you have taken the above steps.”
So quite a change in tone. The MHRA seems to have recognised that contrary to its declared mission ‘to improve health’, its original statement actually endangered the health of tens of thousands of people.
Additionally, solicitors representing the UKCTA have now written to the MHRA seeking clarification of its intentions and making three crucial points:
- The letters to CBD suppliers and the MHRA’s original press statement have caused serious financial damage to the CBD industry.
- The MHRA has conducted no effective consultation with stakeholders.
- The MHRA’s own guidelines require it to consider each product on a case by case basis and a blanket ban on products containing CBD would be unlawful.
Professor Mike Barnes, scientific and medical advisor to CLEAR, commented:
“The MHRA’s new stance is an improvement from their previous position. However, I cannot see any value in delaying only a few months. Some patients might be able to find an alternative medicine from their doctor but many people will have already tried alternative medications and found that CBD is the only satisfactory treatment for their condition. This is the case, for example, for children with epilepsy who will have almost certainly have been under the care of a specialist and tried available anticonvulsants and found that CBD is the only treatment that works for them. The MHRA does not seem to realise the impact of this arbitrary and rushed decision which will clearly be detrimental and potentially have very serious (and in some cases life threatening) implications for some people. The MHRA need to work with the manufacturers and the medical profession to determine the best way forward that both recognises that cannabis based products have medicinal value, and as such need proper trials of efficacy and safety, yet on the other hand does not place existing users at risk of harm”.
MHRA Confirms Meeting With CBD Industry Representatives.
Today, the Medicines and Healthcare products Regulatory Agency (MHRA), has arranged a meeting with representatives from the UK Cannabis Trade Association (UKCTA) to discuss its designation of cannabidiol (CBD) as a medicine.
 A request for a meeting was was first made in writing on 20th September 2016, when the possibility of the MHRA’s action was still little more than a rumour. Nearly six weeks later, after repeated requests, complaints and lobbying from many companies, individuals and MPs, the meeting has been fixed for 3rd November 2016.
A request for a meeting was was first made in writing on 20th September 2016, when the possibility of the MHRA’s action was still little more than a rumour. Nearly six weeks later, after repeated requests, complaints and lobbying from many companies, individuals and MPs, the meeting has been fixed for 3rd November 2016.
The main aim of the meeting will be to discuss interim arrangements for people currently using CBD as a food supplement. Clearly, we will also address concerns over the impact of this decision on many small businesses and the people they employ.
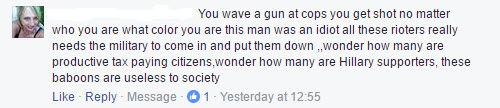
Facebook Says Calling A Black Man A Baboon “Doesn’t Violate Our Community Standards”.
On the other hand Facebook says that recommending a responsible, reputable supplier of verified, lab-tested, legal CBD food supplements does violate its standards.
At a guess (because you can’t get a straight answer from Facebook about anything), the issue is “We prohibit any attempts by unauthorised dealers to purchase, sell or trade prescription drugs, marijuana, firearms or ammunition.”
Now CBD food supplements are fully legal products. They are not prescription drugs. True, CBD is present in cannabis but it is also found in many other plants. So it’s difficult to understand what the problem is – but not as difficult as getting a coherent answer from Mr Zuckerberg and his disciples.
For the ‘offence’ of recommending a CBD supplier your page gets a seriously heavy warning to all page admins, a threat of permanent deletion and I, as the author of the post sharing a link to CBD Oils UK, was banned from Facebook for 30 days. Such is the reality of living under the diktat of the unaccountable, overbearing, bureaucratic monolith that Facebook has become.
However, when some vile American Trumpoid leaves a comment on the CLEAR page calling a black man a baboon, that’s just fine and dandy.
It is time that Facebook was placed under serious regulation for its unfair and oppressive trading practices. It has become so ubiquitous that it now has a responsibility that goes beyond any independent business. It is virtually impossible for individuals and small businesses to operate without a Facebook account. It should be subject to strict standards and forced to comply with fair practices.
I’m all for free enterprise but it’s time to slam Facebook hard for its tax dodging, its failure to take responsibility for publishing abuse and its unfair treatment of users and advertisers.