Posts Tagged ‘vape pen’
CLEAR and GroGlo Establish First UK Clinical Trials on Cannabis for Chronic Pain.
CLEAR has formed a partnership with the research arm of GroGlo, a UK-based manufacturer of high power, LED, horticultural grow lighting.
The plan is to grow cannabis under a Home Office licence for the production of cannabis oil, both as a dietary supplement and for the development of medical products. To begin with, a low-THC crop of industrial hemp will be planted. We will be using the finola strain, originally developed in Finland and known for its short stature and early flowering. Unlike hemp grown for fibre, finola is usually grown for seed and only reaches a height of 160 – 180 cm but we will be removing male plants before they produce pollen and cultivating the female plants to produce the maximum yield of oil from their flowering tops.
The low-THC oil will be marketed as a dietary supplement, commonly known as CBD oil. There is already a burgeoning market in the UK for CBD products, all of which is currently imported from Europe or the USA. In the USA, the CBD products market was said to be worth $85 million in 2015 so there is huge potential here at home. Aside from the benefit of being UK grown and processed, we anticipate achieving a CBD concentration of about 40%, which is higher than most products already on the market.
Cultivation will be in glasshouses supplemented with LED lighting. GroGlo already has an established glasshouse facility in the east of England. Initial trials will experiment with adjusting the LED technology to provide a changing blend of light wavelengths at different stages of plant growth. This is GroGlo’s area of expertise -combining LED lighting and plant sciences, including existing relationships with some of Europe’s top universities. Professor Mick Fuller, GroGlo’s director of plant science, will lead this research and development process.
During the R&D phase, CO2 extraction of oil will be carried out under laboratory conditions at universities in York and Nottingham which already have extensive experience of the process. Each crop will be measured for yield, cannabinoid and terpene content using high pressure liquid chromatography (HPLC). Safety testing will also look for the presence of heavy metals and other contaminants. The results of testing will be fed back into cultivation and extraction processes to maximise yield and quality.
It is anticipated that the first batches of low-THC oil will be ready for market in six months. We are already in discussions with potential distributors and wholesalers. The CBD market in the UK is ripe for an effective marketing campaign which could build a very substantial business for whoever gets it right.
Once we are successfully achieving our production goals with low-THC cannabis, the same testing and development process will begin with high-THC varieties of cannabis. The aim will be to produce a range of oils extracted from single strains, selectively bred and stabilised for different THC:CBD ratios.
Professor Fuller says that GroGlo lighting products “are in use worldwide to grow a range of crops, but some 60% of sales currently come from overseas users growing cannabis for legitimate medical use.” He explains that there is an emerging market for all sorts of nutritional and medicinal plant products but cannabis shows particular promise. GW Pharmaceuticals is the only UK company to enter this market and it has become a world leader, despite the current restrictive legislation. He says: “Together with CLEAR we believe we can help bring a range of safe, high quality UK-produced cannabis products to market within a matter of two to three years.”
A key issue in the development of a successful medicinal cannabis product is the method of delivery. Smoking is not an acceptable solution as inhaling the products of combustion is an unhealthy practice but one of the great benefits of cannabis smoked as medicine is very accurate self-titration. That is the effects of inhaled cannabis are felt almost instantly and so the patient knows when they have taken enough or when they need more to achieve the required analgesic effect.
The oral mucosal spray developed for Sativex is unpopular with patients, many complain of mouth sores from its use and it was developed at least as much with the objective of deterring ‘recreational’ use of the product as with delivering the medicine effectively. It strangles the therapeutic benefits of the cannabis oil of which Sativex is composed in order to comply with the concerns of the medicines regulators about ‘diversion’ of the product into what they would term ‘misuse’. Absorption of the oil is quicker through the mucous membranes of the inside of the mouth than through the gastrointestinal system but, inevitably, some of the oil is swallowed and the pharmacology of cannabis when processed through the gut and the liver is very different.
We believe the best option is a vapouriser device and our intention is to source a ‘vape pen’ of sufficient quality to operate within clinical standards of consistency and safety. Vapourising cannabis oil avoids inhaling the products of combustion but still enables accurate self-titration of dose. A vape pen would provide a handy, convenient and very effective method of consuming medicinal cannabis. However, aside from the technology itself, initial research shows that vapour is more effectively produced when the oil is blended with either vegetable glycerin (VG) or propylene glycol (PG). Establishing the correct ratio of VG or PG to the oil is another important task.
We anticipate that clinical trials for the use of cannabis oil in treating chronic pain could start within two years. We want to compare different oils, ranging from high-CBD to equal ratios of THC:CBD and high-THC content. Prior to that we have to overcome the challenges of cultivation, oil extraction, vapouriser development and assemble the necessary research team and gain ethical approval for the trials. Recruitment for the trials will start in about 18 months time. If you wish to be considered please email ‘paintrials@clear-uk.org’ with brief details of your condition (no more than 100 words). Do not expect to hear anything for at least 12 months but your details will be passed to the research team as a potential candidate.
CLEAR is promoting this venture simply because someone needs to do something to make this happen. For all the campaigning and lobbying of MPs and ministers, at the end of the day, the plants have to be grown and the various legislative hoops have to be jumped through. We cannot wait any longer for a radical change in the law. We have to progress through the government’s regulatory regime if we want to bring real therapeutic benfit to patients.
This opportunity arises because of the vision of GroGlo’s managing director, Mike Harlington and the team of experts he has built around him. There is huge demand for legitimate medicinal cannabis products in the UK which is only going to increase with the inevitable progress towards law reform and increasing awareness of the benefits of cannabis. Together, CLEAR and GroGlo are bringing the great hope that medicinal cannabis offers closer to reality than ever before.
This Is The Future Of Cannabis. For Medicine, Nutrition And Pleasure.
One of these vape pens contains Blue Dream sativa cannabis oil, 91% THC, the other is Hindu Kush indica cannabis oil, 85% THC and the spare cartridge has the dregs of some New York City Diesel sativa, 85% THC. You can’t tell which is which to look at them but each has a distinctive flavour and effect. They’re not completely odour free but almost.
This is the future of cannabis as a consumer product. It is cleaner, neater, handier, healthier and better for you than raw herbal cannabis. Most importantly, for medicinal applications, it homogenises all the compounds into an oil of consistent quality and content meaning that dosage and effect at last becomes predictable and reliable.
I have been investigating this theory for some time but my recent trip to Colorado enabled me to conduct some practical experiments and more thoroughly understand how this idea can work. I am now convinced that this is the way forward for the cannabis industry. Once we achieve legalisation in the UK, which is inevitable, probably in about five years, these pens are how cannabis will become available as a consumer product on the high street. They are also how medicinal cannabis will be dispensed. Your doctor’s prescription will be fulfilled by a cartridge with the appropriate blend of cannabinoids which you screw onto your battery and use immediately. Batteries will also be supplied on prescription, in the same way that syringes or blood glucose meters are for diabetics.
In Colorado dispensaries these pens are already available in a choice of strains and blends. Currently, the popular products contain 250 mg of THC in a blend of cannabis oil and propylene glycol (PG), just as e-cigs contain a nicotine oil and PG.
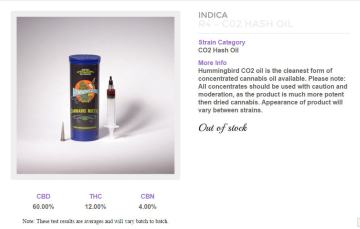
Alternatively, you can buy the oil of your choice and fill the cartridges yourself. This is undoubtedly the way to do it and a wide choice of oils is available, made by CO2 and solvent extraction processes. The Farm, my favourite dispensary in Boulder, is already supplying cannabinoid blends such as a 60% CBD, 12% THC, 4% CBN product which is clearly for medicinal use. I have no doubt that soon we will see a Charlotte’s Web product and Sativex-like blends with equal ratios of THC:CBD. Other, more sophisticated blends of other cannabinoids and probably terpenes will soon follow.
However, I am certain that some propylene glycol is a good thing. The oil vapes much better when diluted and PG is nothing to worry about, it is in many health, cosmetic and food products. It has many uses. It’s a solvent, humectant (keeps things moist), preservative and it helps absorption of some products. It is non-toxic.
There is further development work to be done. I believe there is a ‘sweet spot’ for the correct amount of PG, probably around 20%. I also think the battery and cartridges can be improved, particularly for medical use. Once this is achieved, a product like this with perhaps a 60:40 THC:CBD ratio should form the basis of an application to the Medicines and Health products Regulatory Agency (MHRA) for a marketing authorisation. It will knock Sativex into a cocked hat. In fact, if GW Pharma aren’t investigating this already then they are failing in their duty to shareholders. I shall certainly be doing all I can to research and facilitate the funding to bring such a product to market.
Yes, this is the future of cannabis. Imagine the packaging, marketing and merchandising opportunities for the recreational market. Understand the overwhelming benefits of this as medicine against the raw, herbal product. Yes, I know some will object and the tired old hippy luddites will say it’s a sell out and many more Big Pharma conspiracy theories will emerge but this is the future. Remember you heard it here first.